BPC-157 Research Peptide Overview and RUO Guide
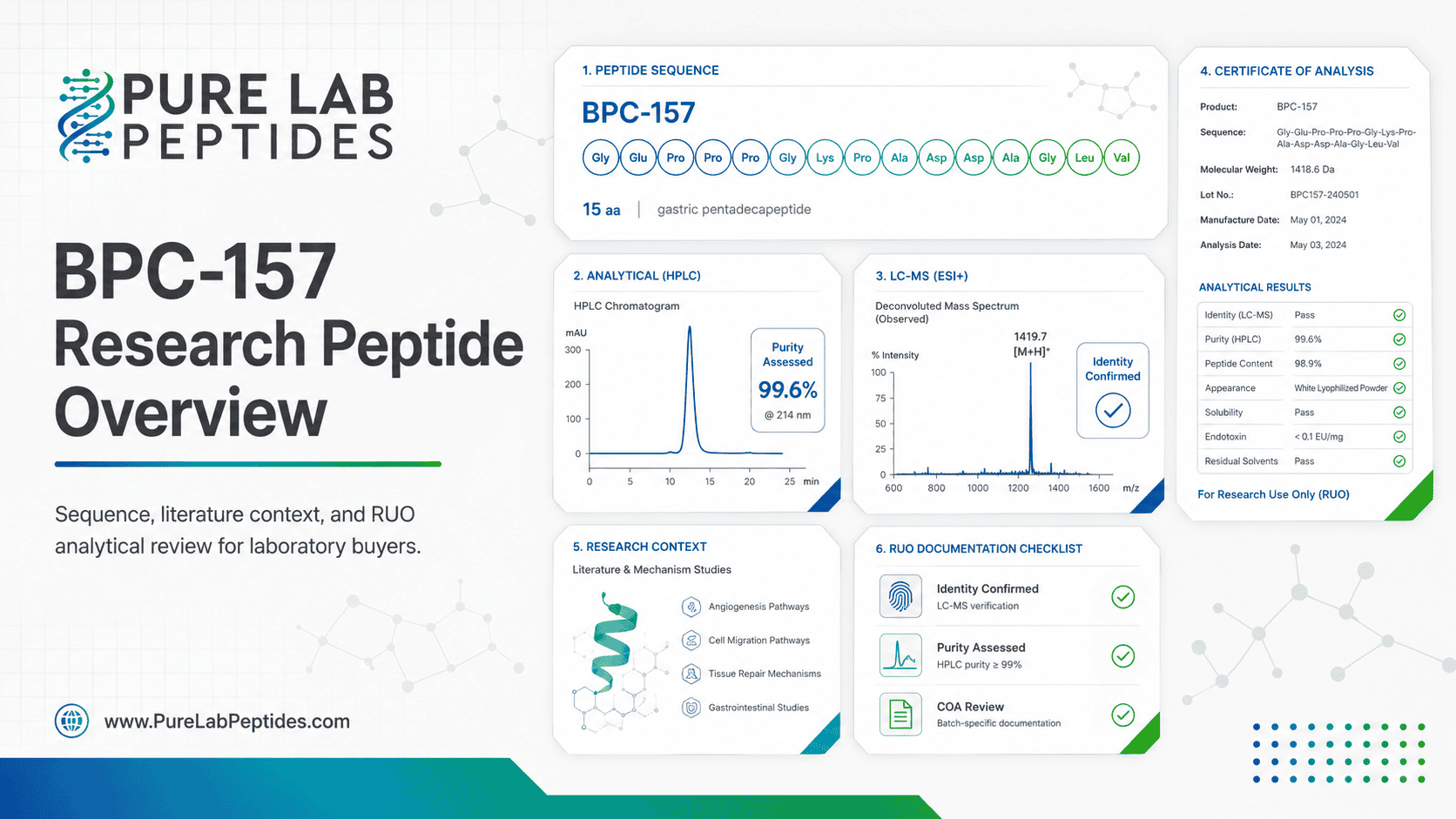
This BPC-157 Research Peptide Overview examines a synthetic pentadecapeptide that appears frequently in preclinical literature on soft-tissue signaling, angiogenesis, and laboratory repair models. For Pure Lab Peptides, the topic is best handled as research-use-only educational content anchored in sequence identity, mechanism studies, analytical documentation, and claim boundaries – not consumer-use narratives. Published literature is substantial, but the evidence base remains dominated by laboratory and nonclinical work.[1][2][3][4]
Fast Answer
BPC-157 is a 15-amino-acid research peptide discussed primarily in preclinical literature, where investigators have examined fibroblast migration, angiogenic signaling, and nitric oxide-related pathways. Products discussed in this article are intended for laboratory research use only and are not intended for human or animal consumption. For research teams and laboratory buyers, the most important questions are identity, salt form, purity, impurity profile, and lot-level documentation.[1][2][5][6][7]
What BPC-157 is in the literature
Published records describe BPC-157 as the pentadecapeptide sequence Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val. PubChem lists the free-base entry as C62H98N16O22, while the FDA UNII record lists synonyms including Bepecin and PL-14736 and notes that UNII availability does not imply regulatory review or approval.[1][2]
That distinction matters because the literature links BPC-157 to body protection compound work associated with human gastric juice, but the material encountered in supplier documentation is synthetic peptide substance and should be described that way. In practice, researchers should verify whether a listing refers to the free base or to a salt form, because PubChem catalogs BPC-157 acetate as a separate substance record rather than treating every label as interchangeable.[2][9]
The EMA guideline on synthetic peptides explains why that level of description is important. Its recommended specifications can include identification, assay/content, total and individual impurities, high-molecular-weight impurities, and counter-ion identity and content, because the counter ion can affect the physicochemical profile of the peptide. For serious laboratory procurement, a BPC-157 document set that omits salt-form detail is missing a core quality variable.[8][9]
Why BPC-157 stays in research focus
BPC-157 remains a recurring topic because the literature connects it to several overlapping experimental themes rather than one narrow question. A 2019 review in Cell and Tissue Research summarized publications across tendon, ligament, muscle, and related soft-tissue models, while a 2025 systematic review in HSS Journal surveyed English-language literature through mid-2024 and showed that the field is still composed mainly of lower-level preclinical evidence.[10][3]
Recent review literature frames BPC-157 as mechanistically active but still unsettled. The 2025 narrative review in Current Reviews in Musculoskeletal Medicine evaluated molecular mechanisms, translational questions, and safety concerns, emphasizing that the compound is widely discussed but not yet supported by a uniformly mature evidence framework beyond laboratory research contexts.[11][3]
For RUO editorial strategy, that means the most accurate educational framing is not “what BPC-157 does” in a consumer sense, but how the literature classifies it: a synthetic pentadecapeptide repeatedly examined in migration, angiogenesis, vascular signaling, and repair-oriented experimental systems. That focus aligns the article with qualified research intent and keeps the discussion close to the actual state of the evidence.[1][2][3][10][11]
Pathways researchers have investigated
No single fully defined receptor explains the complete BPC-157 literature. Instead, published studies point to a cluster of pathway-level observations that recur across fibroblast, endothelial, and vascular models, which is one reason the compound is often described as pleiotropic rather than receptor-simple.[5][6][11]
Fibroblast migration and adhesion signaling
One of the most cited mechanistic papers is the 2011 Journal of Applied Physiology study by Chang and colleagues. In tendon explant and fibroblast experiments, the authors reported increased outgrowth, increased migration, improved survival under oxidative stress conditions, and dose-dependent phosphorylation of FAK and paxillin. Those findings help explain why BPC-157 is so often discussed in repair-related cell biology.[12]
Growth factor-related signaling
The same research group later reported increased growth hormone receptor expression in tendon fibroblasts exposed to BPC-157. That result did not establish a singular receptor identity for the peptide itself, but it did suggest that the compound may be studied as a regulator of existing signaling programs rather than as a simple structural additive.[13]
Angiogenic signaling
In a 2017 Journal of Molecular Medicine paper, Hsieh and colleagues linked BPC-157 to increased VEGFR2 expression and internalization with downstream VEGFR2-Akt-eNOS activation. That paper is repeatedly cited because it ties BPC-157 to a concrete angiogenic signaling axis and provides one of the clearest bridges between cell assays and broader vascular observations in the research record.[5]
Nitric oxide and vasomotor tone
A later study from the same group examined isolated aorta and endothelial systems and reported nitric oxide-dependent vasomotor effects associated with Src-Caveolin-1-eNOS signaling. Together with the VEGFR2 work, this places nitric oxide modulation among the most consistent mechanistic themes in the modern BPC-157 literature.[6]
Taken together, these publications suggest that BPC-157 is best described as a multi-pathway research compound with recurring signals in cell migration, angiogenesis, and endothelial regulation. They do not, however, remove the need for careful interpretation, because pathway observations from isolated or highly specific preclinical systems do not automatically generalize across every research setting.[5][6][11][3]
What the evidence base currently looks like
The evidence base is broader than a single mechanism paper but narrower than many web summaries suggest. The most recent review-level sources indicate that BPC-157 remains primarily a preclinical topic, while dedicated toxicology and ADME papers provide additional context on experimental characterization rather than a final answer on evidence maturity. In other words, the dossier is real, but it is still weighted toward laboratory systems and nonclinical interpretation.[3][11][14][15]
| Study focus | Experimental context | Reported observation | Research takeaway |
| Fibroblast migration and tendon outgrowth | Ex vivo tendon explants and cultured fibroblasts | Increased outgrowth and migration, with FAK-paxillin activation and improved stress survival.[12] | Useful mechanistic signal for connective-tissue research, but still highly model-specific. |
| Growth hormone receptor expression | Cultured tendon fibroblasts | Growth hormone receptor expression increased after BPC-157 exposure.[13] | Suggests interaction with signaling networks rather than a fully mapped primary receptor. |
| VEGFR2 and angiogenic signaling | Endothelial assays and ischemia-associated experiments | VEGFR2 up-regulation and VEGFR2-Akt-eNOS pathway activation were reported.[5] | One of the strongest pathway anchors in the literature, but not a complete mechanism map. |
| Nitric oxide and vasomotor tone | Isolated aorta and endothelial signaling systems | Nitric oxide-dependent vasomotor changes with Src-Caveolin-1-eNOS signaling were described.[6] | Supports the recurrent vascular-signaling theme seen across multiple BPC-157 papers. |
| Preclinical safety package | Nonclinical toxicity studies | The toxicology report described a preclinical safety evaluation program under study conditions.[14] | Important background document, but nonclinical safety work does not equal broad evidence consensus. |
| Pharmacokinetics and disposition | Nonclinical ADME work | A dedicated 2022 study characterized pharmacokinetics, distribution, metabolism, and excretion profiles.[15] | Useful for research context and compound characterization, not a substitute for broader confirmatory literature. |
A useful way to read this table is to separate pathway consistency from evidence maturity. BPC-157 has recurring mechanistic motifs across fibroblast and vascular studies, but the field still depends heavily on experimental systems, review-level synthesis, and a relatively concentrated body of preclinical literature.[3][11][10]
Documentation, analytical testing, and lot review
For laboratory procurement, literature review is only half the task. The other half is whether the material under review is chemically and analytically described with enough precision for the intended project. EMA treats purity as a critical quality attribute for synthetic peptides and recommends specifications that may include identity, total and individual impurities, assay/content, counter-ion identity and content, residual solvents, endotoxins, and microbiological purity.[8]
The same guideline offers a practical analytical roadmap. Mass spectrometry and LC-MS are presented as tools for molecular-mass and sequence confirmation, while LC and ion chromatography can be used for counter-ion assessment. EMA also recommends an orthogonal purity strategy using size-based, charge-based, and hydrophobicity-based separations when needed to represent the full impurity profile of a synthetic peptide.[8]
That is especially relevant for BPC-157 because free-base and acetate records are distinct, and the counter-ion profile can materially change what a label means. If a product listing says “BPC-157” but the analytical documentation reflects acetate content, the documentation should say so directly rather than leaving the reader to infer which version was actually analyzed.[9][8]
Analytical validation is part of the same quality discussion. ICH Q2(R2) states that analytical procedures for identity, purity, impurity, and related measurements should be validated to show they are fit for the intended purpose, while ICH Q1A(R2) explains that stability testing is meant to show how quality changes over time and to help identify degradation pathways under stress conditions. Although these are medicines-focused quality references, they remain useful benchmarks when interpreting peptide COAs and stability claims.[16][17]
The diagram below is an editorial synthesis of a peptide-evaluation workflow rather than a published dataset.
If documentation is thin, the research risk is not just administrative. FDA specifically notes that compounded BPC-157 raises concerns around peptide-related impurities, immunogenicity risk for certain routes, and active-pharmaceutical-ingredient characterization complexity. Those same quality themes are why RUO buyers should read beyond headline purity percentages and look for orthogonal evidence of identity and impurity control.[7][8]
RUO claim boundaries and status context
BPC-157 requires especially careful claim discipline because it sits at the intersection of strong online hype, substantial preclinical literature, and clear regulatory boundary signals. OPSS describes BPC-157 as a synthetic 15-amino-acid peptide, states that it is not a dietary ingredient, and notes that the FDA approved-drugs database does not include it. WADA separately identifies BPC-157 within the Prohibited List framework for sport.[4][18]
For Pure Lab Peptides, the compliant editorial approach is therefore straightforward: describe what the published literature has examined, name the pathways and analytical expectations precisely, and avoid consumer-facing outcome language. In practice, that means centering educational content on literature scope, identity records, COA elements, impurity control, and lot-level transparency rather than on personal-use narratives or outcome claims.[4][7][8][16][17]
FAQs
What is BPC-157 in research literature?
In research literature, BPC-157 is generally described as a synthetic pentadecapeptide associated with body protection compound work and cataloged in public chemical identity databases. Most publications discuss it in the context of laboratory and nonclinical pathway studies rather than as a mature, consensus-standardized research tool with universally agreed endpoints.[1][2][10][3]
Is BPC-157 a naturally occurring peptide or a synthetic research material?
BPC-157 is best described for supplier and documentation purposes as a synthetic research peptide. The literature links its sequence to gastric juice-derived body protection compound research, but the substance encountered in catalog and COA contexts is synthetic material, and the record may need to distinguish the free base from acetate or other counter-ion forms.[2][1][9]
Which pathways are most often discussed in BPC-157 studies?
The pathways most often discussed in BPC-157 studies include fibroblast migration and FAK-paxillin signaling, growth hormone receptor expression in tendon fibroblasts, VEGFR2-Akt-eNOS angiogenic signaling, and nitric oxide-related endothelial regulation through Src-Caveolin-1-eNOS. Those themes recur across several of the most cited mechanistic papers in the field.[12][13][5][6]
What should a BPC-157 COA include?
A BPC-157 COA should make the named substance unambiguous and support it with validated analytical context. At minimum, the documentation should clearly address identity, assay/content, total and individual impurities, and any relevant counter-ion form, while the supporting methods should be fit for their intended identity, impurity, and stability uses.[8][16][17][9]
Why is RUO positioning important for BPC-157?
RUO positioning is important for BPC-157 because the compound is heavily discussed online while the strongest evidence still comes from preclinical literature and regulators have drawn clear boundary lines. Official sources describe it as unapproved, and FDA has separately highlighted impurity and characterization concerns in compounding discussions, which makes precise research-only framing essential.[3][4][7][18]
Next Steps
Review batch-specific documentation before selecting any research-use-only peptide. Explore Pure Lab Peptides for RUO peptide compounds with clear labeling, research-focused product information, and available documentation. For research teams comparing peptide suppliers, prioritize COA availability, transparent labeling, and lot-level documentation.
References
- PubChem. “Bpc-157.” PubChem Compound Summary. 2026. https://pubchem.ncbi.nlm.nih.gov/compound/9941957
- U.S. Food and Drug Administration. “BPC-157.” UNII Search Service. 2026. https://precision.fda.gov/uniisearch/srs/unii/8ED8NXK95P
- Vasireddi N, Hahamyan H, Salata MJ, et al. “Emerging Use of BPC-157 in Orthopaedic Sports Medicine: A Systematic Review.” HSS Journal. 2025. https://doi.org/10.1177/15563316251355551
- Operation Supplement Safety. “BPC-157: A prohibited peptide and an unapproved drug found in health and wellness products.” OPSS. 2025. https://www.opss.org/article/bpc-157-prohibited-peptide-and-unapproved-drug-found-health-and-wellness-products
- Hsieh MJ, Liu HT, Wang CN, et al. “Therapeutic potential of pro-angiogenic BPC157 is associated with VEGFR2 activation and up-regulation.” Journal of Molecular Medicine. 2017. https://doi.org/10.1007/s00109-016-1488-y
- Hsieh MJ, Lee CH, Chueh HY, et al. “Modulatory effects of BPC 157 on vasomotor tone and the activation of Src-Caveolin-1-endothelial nitric oxide synthase pathway.” Scientific Reports. 2020. https://doi.org/10.1038/s41598-020-74022-y
- U.S. Food and Drug Administration. “Certain Bulk Drug Substances for Use in Compounding That May Present Significant Safety Risks.” FDA. 2026. https://www.fda.gov/drugs/human-drug-compounding/certain-bulk-drug-substances-use-compounding-may-present-significant-safety-risks
- European Medicines Agency. “Guideline on the Development and Manufacture of Synthetic Peptides.” EMA. 2025. https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-development-manufacture-synthetic-peptides_en.pdf
- PubChem. “BPC-157 acetate.” PubChem Compound Summary. 2026. https://pubchem.ncbi.nlm.nih.gov/compound/BPC-157-acetate
- Gwyer D, Wragg NM, Wilson SL. “Gastric pentadecapeptide body protection compound BPC 157 and its role in accelerating musculoskeletal soft tissue healing.” Cell and Tissue Research. 2019. https://doi.org/10.1007/s00441-019-03016-8
- McGuire FP, Martinez R, Lenz A, Skinner L, Cushman DM. “Regeneration or Risk? A Narrative Review of BPC-157 for Musculoskeletal Healing.” Current Reviews in Musculoskeletal Medicine. 2025. https://doi.org/10.1007/s12178-025-09990-7
- Chang CH, Tsai WC, Lin MS, Hsu YH, Pang JHS. “The promoting effect of pentadecapeptide BPC 157 on tendon healing involves tendon outgrowth, cell survival, and cell migration.” Journal of Applied Physiology. 2011. https://doi.org/10.1152/japplphysiol.00945.2010
- Chang CH, Tsai WC, Hsu YH, Pang JHS. “Pentadecapeptide BPC 157 Enhances the Growth Hormone Receptor Expression in Tendon Fibroblasts.” Molecules. 2014. https://doi.org/10.3390/molecules191119066
- Xu C, Sun L, Ren F, et al. “Preclinical safety evaluation of body protective compound-157, a potential drug for treating various wounds.” Regulatory Toxicology and Pharmacology. 2020. https://doi.org/10.1016/j.yrtph.2020.104665
- He L, Feng D, Guo H, et al. “Pharmacokinetics, distribution, metabolism, and excretion of body-protective compound 157, a potential drug for treating various wounds, in rats and dogs.” Frontiers in Pharmacology. 2022. https://doi.org/10.3389/fphar.2022.1026182
- International Council for Harmonisation. “ICH Q2(R2): Validation of Analytical Procedures.” ICH Guideline. 2023. https://database.ich.org/sites/default/files/ICH_Q2%28R2%29_Guideline_2023_1130.pdf
- International Council for Harmonisation. “Q1A(R2) Stability Testing of New Drug Substances and Products.” ICH Guideline. 2003. https://database.ich.org/sites/default/files/Q1A%28R2%29%20Guideline.pdf
- World Anti-Doping Agency. “The Prohibited List.” WADA. 2026. https://www.wada-ama.org/en/prohibited-list











