 Save 13%GLP-1 / Incretin Class AnalogsGLP3-R 20mg
Save 13%GLP-1 / Incretin Class AnalogsGLP3-R 20mg
Place your order before 12 PM EST for same-day dispatch.
Enjoy complimentary shipping on orders exceeding $200.
Our dedication to service defines every interaction.
We provide peptides and proteins with over 99% purity.
Featured
 Save 13%GLP-1 / Incretin Class AnalogsGLP3-R 20mg
Save 13%GLP-1 / Incretin Class AnalogsGLP3-R 20mg Peptide Blends & Multi-Component FormulationsKLOW (TB-500 10mg + BPC-157 10mg + KPV 10mg + GHK-Cu 50mg)$199.99
Peptide Blends & Multi-Component FormulationsKLOW (TB-500 10mg + BPC-157 10mg + KPV 10mg + GHK-Cu 50mg)$199.99 Save 35%Peptide Blends & Multi-Component FormulationsBPC-157 + TB-500 (20mg Blend)
Save 35%Peptide Blends & Multi-Component FormulationsBPC-157 + TB-500 (20mg Blend) Neuropeptide & CNS-Active CompoundsSemax 10mg$24.99
Neuropeptide & CNS-Active CompoundsSemax 10mg$24.99 Save 17%Neuropeptide & CNS-Active CompoundsSelank 10mg
Save 17%Neuropeptide & CNS-Active CompoundsSelank 10mg Out of StockMitochondrial-Targeted PeptidesSS-31 30mg$149.99
Out of StockMitochondrial-Targeted PeptidesSS-31 30mg$149.99 Peptide Blends & Multi-Component FormulationsCJC-1295 NO DAC + Ipamorelin (10mg Blend)$84.99
Peptide Blends & Multi-Component FormulationsCJC-1295 NO DAC + Ipamorelin (10mg Blend)$84.99 Melanocortin Receptor LigandsPT-141 10mg$44.99
Melanocortin Receptor LigandsPT-141 10mg$44.99 Peptide Blends & Multi-Component FormulationsGlow (GHK-Cu 50mg + TB-500 10mg + BPC-157 10mg) 70mg Blend$169.99
Peptide Blends & Multi-Component FormulationsGlow (GHK-Cu 50mg + TB-500 10mg + BPC-157 10mg) 70mg Blend$169.99 Mitochondrial-Targeted PeptidesMOTS-C 20mg$199.99
Mitochondrial-Targeted PeptidesMOTS-C 20mg$199.99 Growth Hormone Secretagogues (GHS)Tesamorelin 20mg$199.99
Growth Hormone Secretagogues (GHS)Tesamorelin 20mg$199.99-600x600.png) Dermatological Research ReagentsTri-Heal(TB-500 25mg + BPC-157 10mg + KPV 10mg)$249.99
Dermatological Research ReagentsTri-Heal(TB-500 25mg + BPC-157 10mg + KPV 10mg)$249.99Press & Media
Pure Lab Peptides and our research catalog have been featured across leading news and science publications.






























Research Classes
Browse our full catalog of research compounds organized by mechanism class. For laboratory research use only.

Incretin class research analogs spanning GLP-1, GLP-2, and GIP receptor families.
Explore
GHS research peptides for somatotropic axis and pulsatility studies.
Explore
Research peptides for cellular protection and tissue repair pathway studies.
Explore
Tripeptide-copper complexes for in-vitro laboratory research applications.
Explore
Short synthetic peptide sequences from the Khavinson research body of work.
Explore
Research peptides for mitochondrial bioenergetic and cellular metabolism studies.
Explore Research compounds for metabolic pathway and adipose tissue studies.
Apoptosis
Research compounds for metabolic pathway and adipose tissue studies.
Apoptosis
 Research compounds for programmed cell death and cellular pathway studies.
Cellular Senescence
Research compounds for programmed cell death and cellular pathway studies.
Cellular Senescence
 Research compounds for senescence and cellular aging in-vitro studies.
Dermatological Reagents
Research compounds for senescence and cellular aging in-vitro studies.
Dermatological Reagents
 Research reagents for in-vitro dermal and integumentary system studies.
Hypothalamic-Pituitary Axis
Research reagents for in-vitro dermal and integumentary system studies.
Hypothalamic-Pituitary Axis
 Research peptides targeting the HPG axis and endocrine signaling pathways.
IGF & Insulin-Class
Research peptides targeting the HPG axis and endocrine signaling pathways.
IGF & Insulin-Class
 Insulin-like growth factor and analog research peptides for in-vitro studies.
Lab Solutions & Consumables
Insulin-like growth factor and analog research peptides for in-vitro studies.
Lab Solutions & Consumables
 Solutions and consumables for peptide reconstitution and research workflows.
Melanocortin Ligands
Solutions and consumables for peptide reconstitution and research workflows.
Melanocortin Ligands
 Melanocortin receptor ligands for in-vitro receptor pharmacology research.
Neuropeptide & CNS-Active
Melanocortin receptor ligands for in-vitro receptor pharmacology research.
Neuropeptide & CNS-Active
 Research compounds for in-vitro neuropeptide and CNS signaling pathway studies.
Oxytocin-Family
Research compounds for in-vitro neuropeptide and CNS signaling pathway studies.
Oxytocin-Family
 Oxytocin and posterior pituitary research peptides for in-vitro studies.
Peptide Blends
Oxytocin and posterior pituitary research peptides for in-vitro studies.
Peptide Blends
 Pre-formulated multi-component research peptide blends for in-vitro studies.
Thymus-Derived
Pre-formulated multi-component research peptide blends for in-vitro studies.
Thymus-Derived
 Thymic and immunomodulating research peptides for in-vitro immune signaling studies.
Thymic and immunomodulating research peptides for in-vitro immune signaling studies.
All compounds with 99%+ purity and third-party COA documentation.
Standards You Can Verify
Every research compound we supply is held to verifiable standards; measurable purity, independent documentation, and a transparent in-vitro research-use framework.

Every batch is synthesized to a minimum 99% purity threshold, verified by HPLC analysis before release.

Every product ships with a certificate of analysis from an independent laboratory — full transparency, every order.

Supplied exclusively for in-vitro laboratory research. Not for human consumption or any in-vivo application.
Research & Education
Evidence-based guides on peptide purity, documentation, and handling — written for RUO laboratory buyers and research teams.

This article reviews how UV/visible light can degrade research peptides and outlines analytical methods and handling practices to preserve peptide integrity in…
Jun 6, 2026 · 10 min read
Copper–peptide complexes are coordination compounds studied in lab research. This article explains their coordination, analytical characterization (MS, EPR), and applications under RUO…
Jun 6, 2026 · 12 min read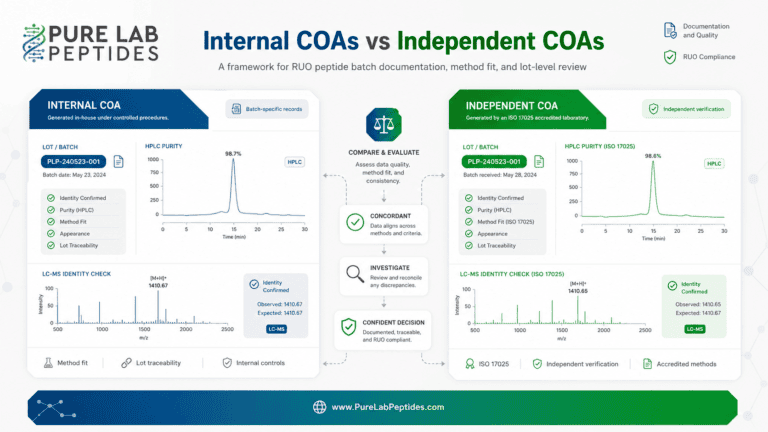
Internal and independent COAs answer different documentation questions in RUO peptide sourcing. This article explains how to compare lot-specific supplier records and…
Jun 6, 2026 · 21 min readIn-depth guides on purity, identity, blends, stability, and handling — updated as new research is published.
Researchers looking to buy peptides online for laboratory research should evaluate peptides as research-use-only laboratory materials, not consumer products. For laboratory buyers, the key considerations are peptide identity, purity documentation, batch-specific COAs, lot traceability, product labeling, analytical testing, product form, storage information, and supplier transparency. This guide explains how to evaluate peptides for controlled research procurement through Pure Lab Peptides while keeping the discussion limited to documentation, quality review, and research-use-only sourcing.
Researchers can buy peptides online for laboratory research by reviewing RUO labeling, batch-specific COA documentation, purity data, identity information, storage guidance, and supplier transparency before selecting a source. Products discussed in this article are intended for laboratory research use only and are not intended for human or animal consumption.
The phrase buy peptides online is addressed here as laboratory research procurement intent, not personal-use intent. In this context, “buy peptides” refers to the process qualified researchers, laboratory buyers, research institutions, and technical procurement teams use to evaluate peptide materials before adding them to a controlled research workflow.
That evaluation should focus on documentation. A research procurement review should ask whether the supplier clearly labels peptide materials for research use only, whether a batch-specific certificate of analysis is available, whether purity and identity information are documented, whether the lot number is traceable, whether the product form is clearly stated, and whether storage and handling information is available before laboratory intake.
Research-use-only sourcing should also be consistent with product labeling and supplier language. FDA guidance discussing products labeled for research use only emphasizes that RUO positioning should align with the product’s stated intended use, which is why peptide procurement language should remain separate from clinical, diagnostic, therapeutic, or consumer-use language [1].
Peptides are generally defined as compounds formed from two or more amino acid residues linked by peptide bonds [2]. IUPAC-IUB nomenclature guidance also provides standardized conventions for representing amino acids and peptide sequences in scientific contexts [3]. In laboratory documentation, peptides may be described by sequence, residue count, molecular weight, terminal groups, salt form, modification pattern, product form, analytical method, and purity documentation.
Because this article discusses peptides as a broad research-material category, the term “peptides” should not be treated as one compound. A research peptide may be a short peptide, a longer synthetic peptide, a peptide fragment, a cyclic peptide, a modified peptide, a peptide analog, or a multi-component peptide blend. PubChem describes peptides as compounds formed from two or more amino acids connected by peptide bonds, while IUPAC separately defines polypeptides as peptides containing ten or more amino acid residues [4] [5].
For procurement purposes, the broad category name is only the starting point. Researchers should evaluate the specific product name, sequence or molecular description where relevant, stated molecular weight where relevant, batch-specific COA, identity testing, purity documentation, lot number, product form, and storage documentation.
Modern peptide research materials are often discussed in connection with synthetic peptide chemistry. Merrifield’s work on solid-phase peptide synthesis established a foundation for sequence-directed peptide synthesis, and later Fmoc-based peptide synthesis literature describes widely used approaches for assembling synthetic peptides [6] [7] [8]. Reaction monitoring methods, including color tests for free terminal amino groups, have also been discussed in solid-phase peptide synthesis workflows [9].
Laboratory buyers search to buy peptides online because peptide procurement often requires more than product availability. Technical teams may need to compare RUO labeling, COA access, lot-level documentation, identity data, purity records, storage information, product form, and supplier transparency before selecting a research-use-only source.
For a broad category such as peptides, this review is especially important. Different peptide materials may differ in sequence, residue count, molecular weight, modifications, analytical behavior, and storage considerations. A product category name alone does not establish whether a specific research material is suitable for a laboratory workflow. Documentation should be reviewed at the product and batch level.
Researchers comparing suppliers should prioritize clear research-use-only positioning, batch-specific COA documentation, HPLC or LC-MS information where provided, lot number consistency, and transparent product-page details. Supplier language should remain focused on compound identity, purity documentation, analytical review, and laboratory procurement rather than outcomes, personal use, or product-use claims.
A peptide catalog may include several types of research materials. Each category can require a slightly different documentation review, but the core procurement questions remain the same: What is the material? How is identity documented? How is purity documented? Is the COA batch-specific? Does the lot number match across the product record and documentation?
| Research Peptide Category | Documentation Focus | Procurement Note |
| Sequence-defined peptides | Sequence, molecular weight, identity method, purity data, and lot number | Review the COA and product page together before laboratory intake. |
| Modified peptides | Modification identity, terminal groups, molecular mass, and analytical method | Confirm that documentation identifies the listed molecular form. |
| Peptide fragments | Fragment identity, residue range, molecular weight, and purity documentation | Compare naming, sequence information, and COA details for consistency. |
| Cyclic peptides | Sequence, topology-related identity details, mass data, and purity | Evaluate identity documentation carefully because structure may affect characterization. |
| Peptide blends | Component identity, component-level documentation, and lot traceability | Blend composition should be evaluated through documentation and identity review, not expected outcomes or use protocols. |
When researchers evaluate Buy Peptides Online options, the strongest quality signals are documentation signals. A peptide supplier should make it easy to review research-use-only positioning, batch-specific COA documentation, identity data, purity information, lot number consistency, product form, and storage guidance. These records help technical buyers evaluate whether the material can be logged into a controlled research workflow.
| Evaluation Area | What Researchers Should Review | Why It Matters for RUO Procurement |
| RUO labeling | Confirm the product is clearly labeled for research use only | Helps separate research procurement from human-use positioning |
| COA availability | Review the batch-specific certificate of analysis | Supports lot-level documentation and quality review |
| Purity data | Look for analytical support for the stated purity | Helps evaluate material consistency |
| Identity testing | Review HPLC, LC-MS, mass spectrometry, or related identity data | Helps confirm the material matches the listed peptide |
| Lot traceability | Match lot numbers across product and documentation | Supports research recordkeeping |
| Product form | Confirm whether the material is supplied as lyophilized powder or another documented form | Supports laboratory planning |
| Storage information | Review storage and handling documentation | Supports material tracking in controlled research settings |
| Supplier language | Confirm the supplier avoids dosing, therapeutic, diagnostic, or personal-use claims | Supports research-use-only positioning |
A peptide COA should be reviewed as a batch-specific record, not a general marketing statement. Researchers should look for the peptide name, product identifier, lot number, test date, stated purity percentage, analytical method, identity confirmation, molecular weight where relevant, sequence or formula where relevant, chromatographic or mass data where provided, product form, and storage documentation.
A purity percentage alone does not establish complete compound identity; researchers should evaluate purity, identity, method, lot number, and documentation together. HPLC literature describes chromatographic methods used for peptide separation and purification, including reversed-phase, size-exclusion, ion-exchange, and related approaches [10]. Additional HPLC methodology literature supports the role of chromatographic analysis in peptide evaluation [11].
Mass spectrometry literature also supports peptide sequencing, mass-based identity confirmation, and broader peptide characterization workflows [12] [13]. Synthetic peptide mass spectrometry literature describes MS as a useful method for evaluating synthetic peptide identity and purity in analytical settings [14].
LC-HRMS and LC-MS literature also describes mass-based approaches for peptide quality control, impurity characterization, and related-substance review [15] [16]. Peptide impurity profiling literature shows why related materials, structurally similar impurities, and method selection can matter in quality review [17].
Independent evaluations of synthetic research peptides have reported that supplier-stated purity and independent analytical findings may not always align, reinforcing the importance of reviewing batch-specific documentation rather than relying on headline purity statements alone [18]. Additional analytical work on peptide impurities, purity estimation, and chiral purity analysis further illustrates why multiple documentation points may be relevant to peptide quality review [19] [20] [21].
Analytical-method documentation helps researchers understand how a peptide batch was evaluated. HPLC documentation may support purity assessment and chromatographic separation review. LC-MS or mass spectrometry documentation may support identity confirmation, molecular-mass review, or impurity investigation. The specific interpretation depends on the peptide, method, and batch-specific documentation.
| Analytical Method | What It Can Support | How Researchers Should Review It |
| HPLC | Purity review, chromatographic separation, peak profile evaluation | Compare stated purity with COA and chromatographic documentation. |
| LC-MS | Identity review, mass confirmation, impurity characterization | Confirm that the listed peptide matches the analytical identity record. |
| Mass spectrometry | Mass-based identity support and structural confirmation support | Use mass data as part of the documentation package, not as a standalone supplier claim. |
| Reference-standard style documentation | Traceability, vialing, analytical testing, and stability documentation | Use reference-standard principles to assess documentation completeness. |
Lot traceability connects the product label, COA, receiving record, and inventory record. A laboratory buyer should be able to compare the product name, lot number, product form, and analytical documentation across those records. When the same lot number appears consistently across product documentation, the laboratory record is easier to verify and maintain.
Reference-standard literature discusses vialing, lyophilization, analytical testing, and stability studies as part of quality-supporting documentation for peptide materials [22]. NIST resources also describe how certificates of analysis and lot identifiers support traceability in reference-material contexts [23] [24].
For peptide procurement, lot-level records help qualified researchers confirm that the COA being reviewed corresponds to the material being received. This is especially important when comparing multiple peptides, multiple batches, or multi-component peptide materials.
Pure Lab Peptides presents peptide materials in lyophilized powder form. For research procurement, product-form language should be paired with documentation review. Researchers should review the product page, batch-specific COA, label information, and storage guidance before adding any research-use-only material to laboratory inventory.
Drying and freeze-drying literature discusses solid-state formats as part of stability-oriented approaches for biopharmaceutical materials [25]. Freeze-drying is also widely discussed in formulation literature involving structurally complex active ingredients and carrier systems [26]. Broader protein and peptide stability literature describes multiple stresses that may affect peptide and protein stability, supporting the importance of storage documentation in laboratory workflows [27].
This section is limited to research documentation. It does not provide preparation, administration, dosing, or use instructions. Storage and handling information should be reviewed through supplier documentation and internal laboratory procedures.
Researchers comparing peptide suppliers should focus on supplier transparency, documentation consistency, analytical review, and research-use-only positioning. A strong supplier record allows technical procurement teams to evaluate peptides without relying on unsupported claims.
| Supplier Signal | Strong Research Procurement Signal | Weak or Risky Signal |
| RUO labeling | Clear research-use-only language across product pages and documentation | Ambiguous language that mixes research materials with personal-use positioning |
| COA access | Batch-specific COA documentation available for review | Generic purity statement without batch-level records |
| Identity testing | HPLC, LC-MS, mass spectrometry, or related analytical method listed | No analytical method stated |
| Lot traceability | Lot number matches the label, COA, and product record | No lot-level documentation trail |
| Product language | Research procurement, compound identity, purity documentation, analytical review | Outcome-based claims, personal-use framing, or protocol-oriented language |
| Product form | Lyophilized powder form and storage documentation clearly stated | Product form or storage information is unclear |
Research-use-only peptide procurement is not the same as regulated drug-product submission. However, analytical documentation principles can still help researchers evaluate whether supplier records are complete, consistent, and technically credible. FDA analytical-method guidance discusses documentation supporting identity, strength, quality, purity, and related analytical attributes in regulated contexts [28].
ICH Q2(R2) provides a framework for analytical procedure validation, and ICH Q14 discusses science- and risk-based approaches for analytical procedure development [29] [30]. ISO/IEC 17025 is also relevant as a general standard for testing and calibration laboratories because it addresses competence and valid results [31].
For research peptide buyers, these sources do not replace the product page or batch-specific COA. They provide background for why identity, purity, method documentation, lot traceability, and recordkeeping matter when comparing peptide suppliers.
Published peptide literature spans nomenclature, synthesis, purification, analytical testing, mass spectrometry, impurity profiling, reference standards, and stability. This literature is useful for understanding how peptides are characterized in scientific and laboratory settings. It should not be interpreted as product-use guidance for research-use-only materials.
Peptide literature may be analytical, database-based, methodological, review-based, in vitro, preclinical, or clinical depending on the specific peptide and research question. Published clinical literature should not be interpreted as use guidance for RUO materials. Regulated medicines, clinical-study materials, and research-use-only materials are not interchangeable.
Because peptides are a broad category, researchers should not extrapolate from one peptide sequence, peptide analog, peptide fragment, or peptide blend to another. Documentation should be evaluated at the product and batch level. Research literature can help contextualize peptide chemistry and analytical review, but it does not replace COA review, identity testing, purity documentation, or lot traceability.
| Research Area | What Literature Examines | Evidence Type | RUO Interpretation |
| Peptide identity | Molecular structure, sequence, formula, residue count, or classification | Database / nomenclature / analytical literature | Supports identification and documentation review, not product-use claims |
| Synthetic peptide context | Solid-phase peptide synthesis, Fmoc chemistry, and reaction monitoring | Methods / review / analytical chemistry | Useful for scientific context, not supplier claims by itself |
| Analytical testing | Purity, identity, impurity profiling, and batch verification | HPLC / LC-MS / mass spectrometry / COA | Supports documentation review |
| Storage and stability | Material form, lyophilized powder, drying technologies, and handling considerations | Laboratory documentation / review literature | Supports research workflow planning |
| Supplier documentation | COA, lot number, product labeling, storage information, and traceability | Procurement documentation / quality systems context | Supports laboratory recordkeeping |
| Research-Safe Statement | Why It Is Acceptable | Non-Compliant Version to Avoid |
| “Peptides are discussed in published research related to peptide chemistry, analytical characterization, and laboratory quality review.” | Describes literature context without making a product claim | “Peptides help with human outcomes.” |
| “Researchers should review COA and identity data before procurement.” | Focuses on documentation and quality review | “Users should buy peptides for results.” |
| “Pure Lab Peptides supplies peptides as research-use-only materials.” | Clarifies intended use | “Pure Lab Peptides supplies peptides for therapy.” |
| “The phrase buy peptides online is addressed as research procurement intent.” | Qualifies buying intent as laboratory procurement | “Buy peptides online for personal use.” |
| “Blend composition should be evaluated through documentation and identity review, not expected outcomes or use protocols.” | Supports multi-component documentation review without outcome claims | “Peptide blends should be selected for combined results.” |
Pure Lab Peptides presents peptides as research-use-only laboratory materials. For technical buyers evaluating peptide procurement, the most relevant quality signals are RUO positioning, a stated ≥99% purity claim, lyophilized powder form, batch-specific COA availability, product page documentation, storage and handling information, lot-level traceability, and supplier transparency.
Because peptides are a broad research-material category, researchers should review the relevant product page or peptide collection page rather than assuming one peptide record applies to every peptide. Review the Pure Lab Peptides research peptide collection for RUO labeling, product details, purity information, and batch-specific documentation.
Technical teams comparing peptide suppliers should prioritize batch-specific records, clear labeling, and documentation consistency over broad category claims. A transparent supplier record should help researchers evaluate product identity, purity documentation, analytical testing, lot traceability, storage guidance, and research-use-only positioning before procurement.
Buy peptides online should not be interpreted as personal-use guidance on this page. The phrase is addressed as laboratory procurement intent for qualified researchers reviewing RUO labeling, documentation, purity data, identity information, product form, storage information, and supplier transparency.
Published literature can provide scientific context about peptide nomenclature, synthesis, purification, analytical testing, impurity profiling, and research categories. It does not convert an RUO peptide into a consumer product, clinical material, diagnostic material, veterinary material, or personal-use item.
Purity percentage is only one part of a documentation review. Researchers should evaluate purity, identity, method, lot number, product name, molecular information, chromatographic data, mass data where provided, and COA consistency together. A high stated purity does not replace identity confirmation.
For research procurement, batch-specific documentation matters because the COA should correspond to the lot being procured or received. Technical buyers should match the product name, lot number, and COA details to support laboratory recordkeeping and traceability.
RUO labeling does not support human or animal consumption. It indicates that the material is intended for controlled laboratory research use only. Supplier language should not blur research-use-only positioning with consumer, clinical, diagnostic, veterinary, or outcome-oriented language.
Supplier claims should not replace COA review, identity testing, purity documentation, or lot traceability. A research procurement decision should be based on product-page consistency, batch-specific records, analytical documentation, product form, storage information, and clear RUO positioning.
Researchers can buy peptides online for laboratory research from suppliers that clearly present RUO labeling, batch-specific COA documentation, purity information, identity testing details, product form, storage guidance, and transparent supplier policies. Pure Lab Peptides positions peptide materials for research use only and provides batch-specific documentation for review.
Before buying peptides online, researchers should check RUO labeling, the batch-specific COA, stated purity, analytical method, identity documentation, lot number consistency, product form, storage information, and supplier language. The supplier should avoid consumer-style claims and should not frame peptides for human or animal consumption.
A COA matters when buying peptides because it supports batch-level documentation. Researchers should review the peptide name, lot number, purity percentage, testing method, identity information, date of analysis, and document consistency. A COA helps procurement teams evaluate whether the material record aligns with laboratory documentation requirements.
Peptides discussed on this page are not intended for human or animal consumption. This article addresses peptide sourcing only as research-use-only laboratory procurement. Qualified researchers should evaluate product labeling, COA documentation, identity testing, purity data, storage information, and supplier transparency within controlled laboratory procurement workflows.
Research use only means peptides are supplied for controlled laboratory research settings and are not positioned for consumer, clinical, diagnostic, veterinary, or personal use. For procurement, RUO status should be supported by supplier language, product labeling, documentation, COA availability, and the absence of outcome-based claims.
Published literature about peptides should be interpreted as scientific context, not product-use guidance. Researchers may review literature on peptide nomenclature, synthesis, HPLC, LC-MS, mass spectrometry, reference standards, stability, and analytical methods. That literature should not be used to infer directions, outcomes, or applications for RUO materials.
For research teams comparing peptide suppliers, prioritize COA availability, transparent RUO labeling, purity documentation, identity testing, product form, storage information, and lot-level traceability. Review the Pure Lab Peptides research peptide collection for RUO product details, purity information, and batch-specific documentation.
Discount Applied Successfully!
Your savings have been added to the cart.

Pure Lab Peptides
You must be at least 21 years of age to enter this site. By entering, you confirm that you are of legal age and agree to our terms.
For research use only.