Peptide Purity vs Peptide Identity for RUO Labs
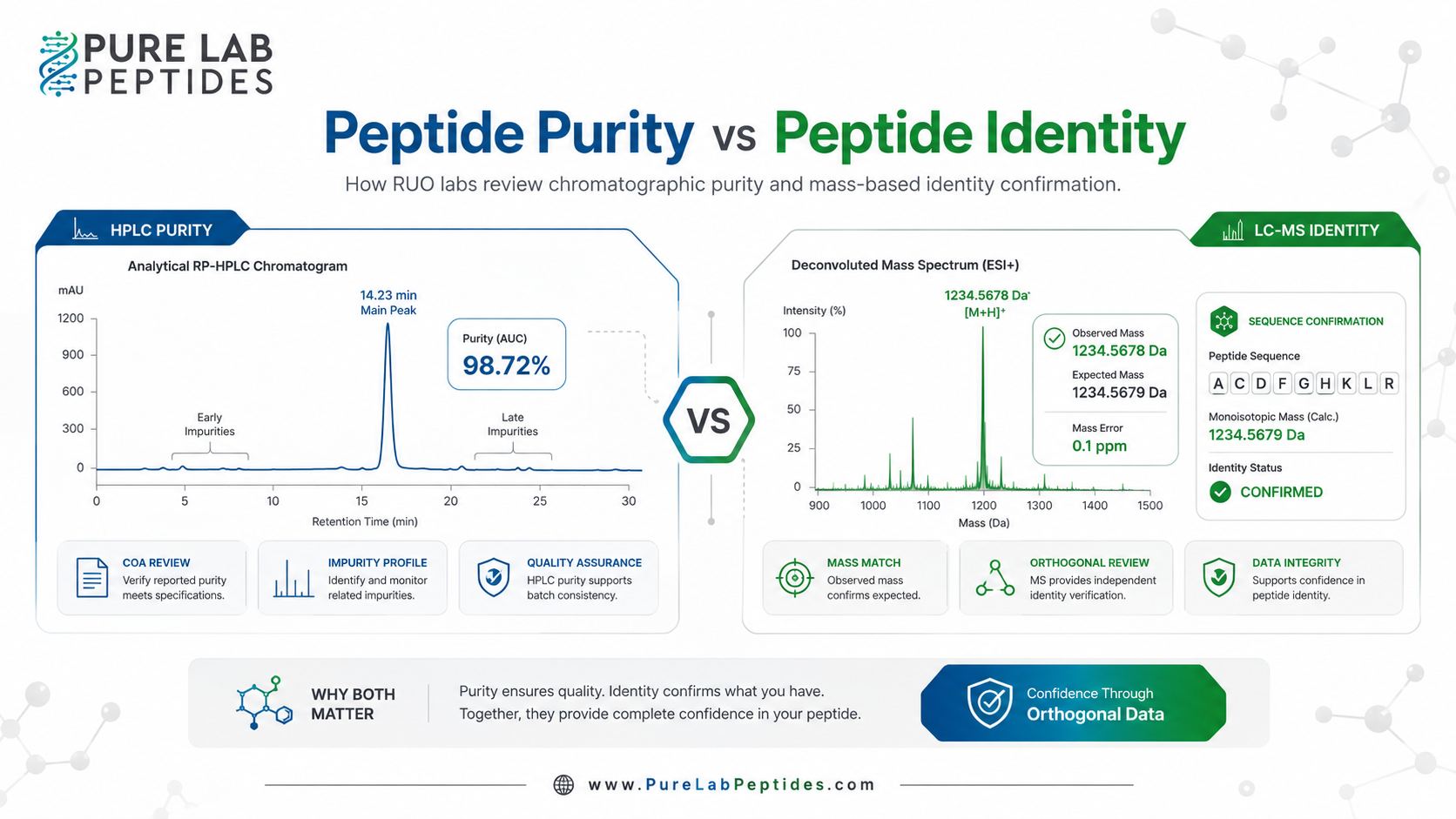
Peptide purity vs peptide identity is one of the most important distinctions in peptide quality review, especially when a laboratory is comparing research-use-only materials across suppliers. Purity addresses how much of a batch is the intended primary component relative to related and unrelated impurities, while identity asks whether the material is actually the peptide it is claimed to be. For RUO sourcing and documentation review, these are separate analytical questions that should not be collapsed into a single number.[1][4]
Fast Answer
Peptide purity and peptide identity are not interchangeable: purity estimates how much of a sample corresponds to the target material, whereas identity verifies that the target material is actually the claimed peptide and not a closely related analog, isomer, or mislabeled compound. Products discussed in this article are intended for laboratory research use only and are not intended for human or animal consumption.[1][2][4]
What Peptide Purity Means
In analytical terms, purity is about the proportion of the primary component relative to secondary components that may interfere with the intended application of the material. IUPAC notes that “pure” is a practical analytical concept, not merely a marketing adjective, and that purity is commonly delivered as a mass fraction of the primary component after relevant secondary components are considered. For peptides, that distinction matters because the number printed on a chromatogram is often narrower than the total composition of the vial.[1][3]
For synthetic peptides, current EMA guidance frames purity as part of a broader specification package that can include total impurities, individual impurities, high molecular weight species such as dimers or aggregates, water content, residual solvents, elemental impurities, and counter-ion content where relevant. In other words, peptide purity is not always a single test. It is often a collection of measurements that describe what else is present besides the intended peptide.[1][4]
That is why a supplier claim such as “99% HPLC purity” should be read carefully. In many peptide workflows, that figure is a chromatographic estimate based on the relative area of the principal peak under a defined LC method and detector setting. It is usually informative for related-impurity profiling, but it does not automatically speak to water, salt form, residual solvents, unresolved co-eluting species, or whether the main peak itself is the correct peptide sequence.[3][9]
What Peptide Identity Means
Peptide identity asks a different question: “Is this actually the claimed peptide?” ICH Q6A states that identification testing should discriminate between closely related structures likely to be present and that a single chromatographic retention time is not regarded as sufficiently specific by itself. The same guideline accepts orthogonal combinations such as two chromatographic procedures based on different principles or combined methods such as HPLC/MS or GC/MS.[4]
The 2025 EMA peptide guideline makes that principle more explicit for synthetic peptides. It recommends at least two orthogonal methods for peptide identification and lists molecular mass, relative retention time, LC-MS, peptide mapping, amino acid analysis, and NMR among the acceptable options. The reason is simple: identity is not just a matter of seeing one large peak, but of unambiguously confirming the peptide sequence and structure that the batch is supposed to represent.[1][4]
Identity can also extend beyond nominal molecular mass. Current peptide guidance and the recent review literature on enantiomeric purity show that stereochemical issues, isobaric substitutions, and closely related sequence variants can require dedicated methods, including chiral chromatography, amino acid analysis, or NMR. A batch can therefore match an expected mass while still raising unresolved identity questions if stereochemistry or sequence-level ambiguity remains.[1][5][10]
How Purity and Identity Diverge in Real Analytical Work
The easiest way to separate the concepts is to think about their failure modes. A peptide can be relatively pure yet fail identity if the dominant component is not the intended sequence. It can also pass an identity check at the level of expected mass or retention behavior while still failing purity because related impurities, degradants, salts, or unresolved co-eluting species remain at unacceptable levels for the intended laboratory work. Identity says what the main thing is; purity says what else is there and in what proportion.[1][4]
The comparison below synthesizes how ICH Q2, ICH Q6A, the EMA synthetic peptide guideline, and peptide reference-standard literature distinguish identity, purity, and closely related quality attributes.[1][2][4][9]
| Attribute | Core question | Common analytical approaches | Typical evidence on a COA | What it does not prove by itself |
| Purity | How much of the batch corresponds to the intended primary component? | RP-HPLC or UPLC, impurity profiling, tests for water, residual solvents, counter-ions, and high molecular weight species when relevant | Main peak percentage, total impurities, individual impurity values, impurity chromatogram | That the main peak is necessarily the correct peptide |
| Identity | Is the batch actually the claimed peptide? | LC-MS, peptide mapping, NMR, amino acid analysis, relative retention time, chiral methods where needed | Observed mass, sequence confirmation, orthogonal identification methods, comparison to a reference standard | That impurity burden is low enough for sensitive laboratory work |
| Assay or content | How much target material is quantitatively present? | Chromatographic assay, amino acid analysis, qNMR, elemental or nitrogen analysis in some workflows | Content value against a reference standard | That impurity distribution and identity have been fully characterized |
Modern peptide literature shows why this distinction persists even in sophisticated LC-MS environments. Work on peak purity in pharmaceutical peptides has emphasized that species with the same mass-to-charge ratio are not readily differentiated by mass spectrometry alone and therefore may still require stronger chromatographic separation to reveal hidden main-peak heterogeneity. That is a purity problem that can survive an apparently straightforward mass confirmation step.[6]
Conversely, LC-HRMS workflows for peptides are powerful because they can connect chromatographic peaks with accurate mass and fragment evidence, helping laboratories determine whether observed impurities are deletions, insertions, oxidized species, or other related structures. That capability strengthens identity assessment and impurity characterization at the same time, but it does not erase the conceptual difference between the two attributes.[7][8]
How Laboratories Evaluate Each Attribute
A serious peptide quality workflow usually begins with fit-for-purpose analytical methods rather than a generic purity label. ICH Q2(R2) defines method validation around intended analytical purpose and shows that specificity is normally evaluated for identity and impurity or purity methods alike, while quantitative impurity methods also depend on characteristics such as accuracy, repeatability, and intermediate precision. The practical implication is that a supplier should be able to explain what a result measures, how it was validated, and what acceptance logic sits behind the number.[2]
For peptide identity, orthogonal confirmation is now standard analytical thinking. The EMA synthetic peptide guideline identifies LC-MS, peptide mapping, amino acid analysis, and NMR as appropriate options, while USP reference-standard work describes multi-method characterization that often combines HPLC retention behavior, MS, NMR, chiral testing, and sequence-specific workflows. In practice, this means identity is strongest when at least two independent analytical principles point to the same conclusion.[1][9]
For purity, laboratories often pair chromatographic separation with orthogonal detection because peptide impurities can be structurally close to the target sequence. LC-HRMS studies and broader LC-MS reviews describe workflows that connect peak-level impurity profiling to accurate-mass assignment and, where needed, sequence-related fragment confirmation. That matters because peptide-related impurities can arise from insertion, deletion, oxidation, glycosylation, incomplete deprotection, or other synthesis- and storage-linked routes, and they are not always fully resolved by a single routine UV trace.[7][8][10]
Illustrative workflow for reviewing peptide purity and identity on a lot-specific certificate of analysis. This diagram is an editorial synthesis rather than a published data figure.[1][2][4][9]
When laboratories need a more compositionally rigorous number, they may move beyond a single chromatogram and use a mass-balance style approach. IUPAC describes purity assignment in terms of the mass fraction of the primary component after accounting for relevant impurity classes, and the USP peptide reference-standard literature describes two-step value assignment strategies that integrate impurities, solvents, moisture, and other non-peptide contributors. This is one reason assay, purity, and identity often appear as separate lines on better peptide documentation.[3][9]
Why Both Matter for Research Quality and Supplier Review
The practical consequences are not hypothetical. In a published quality-control study of 98 synthetic quorum sensing peptides that were requested with at least 95% purity, only 44% met that target under in-house evaluation, and one sample had a main compound with a structure different from the intended peptide. That single paper neatly demonstrates both sides of the problem: purity claims can be overstated, and identity errors can survive into supplied material.[11]
Commercial peptide impurity issues have also been documented in other settings. Currier and colleagues reported that impurities in commercial synthetic peptides could materially affect assay interpretation, reinforcing the broader lesson that peptide lots are analytical inputs whose hidden heterogeneity can alter experimental signal. For research groups, that is a sourcing and documentation issue, not just an abstract quality-theory issue.[12]
That is why batch-specific documentation matters so much. ICH Q7 states that authentic certificates of analysis should be issued for each batch on request and that the certificate should list each test performed, the acceptance limits, and the numerical results obtained when results are numerical. A useful peptide COA therefore links a lot number to actual analytical work. A generic template or recycled chromatogram does not do that.[13]
Reference standards are part of the same story. USP-focused work on peptide standards explains that assigned values for identity, purity, and content are typically built from multiple orthogonal methods and, where needed, multiple laboratories. For supplier comparison, that means stronger documentation usually comes from a system that treats peptide identity and peptide purity as independent but connected analytical attributes rather than as interchangeable marketing language.[9]
Common Misreadings of Peptide COAs
Does a high HPLC percentage mean the peptide is fully characterized?
No. A high HPLC percentage usually indicates favorable chromatographic purity under one defined method, but full characterization can require additional information about identity, water, counter-ions, residual solvents, high molecular weight impurities, and unresolved or poorly separated related species. In other words, a strong HPLC number is useful evidence, but it is not the whole analytical picture.[1][3][9]
Does a correct mass confirm the batch is analytically clean?
Not by itself. Mass confirmation can strongly support peptide identity, but it does not automatically quantify the full impurity burden. Published peptide guidance and LC-MS literature both show that insertion, deletion, oxidation, stereochemical variation, and other related impurities may still need dedicated impurity profiling and orthogonal confirmation, especially when closely related species are expected from synthesis or storage pathways.[1][7][8][10]
Can one main chromatographic peak still hide a problem?
Yes. Peptide-specific guidance now explicitly warns about the risk of co-eluting impurities, and 2D-LC-MS work on peptide peak purity has focused on the fact that same-mass species may not be distinguishable by MS alone. A dominant main peak is encouraging, but it is not unassailable proof that the peak contains only the intended peptide and nothing closely related.[1][6]
Is a generic certificate enough if the method looks familiar?
No. ICH Q7 expects authentic batch-specific certificates that identify the material, the batch, the tests performed, the limits, and the numerical results. A familiar-looking chromatogram without lot traceability, acceptance criteria, release details, or orthogonal identity evidence is weak documentation for research purchasing and should be treated cautiously.[13]
At minimum, a research team comparing peptide lots should expect a lot number tied to the analytical result set, named methods, quantitative result values, impurity information, and some form of orthogonal identity evidence when sequence-level ambiguity is plausible. When those elements are absent, the document is better read as commercial support material than as a strong analytical record.[1][13]
FAQs
Can a peptide show high purity and still fail identity?
Yes. A peptide can show a dominant chromatographic peak and still fail identity if that dominant component is not the intended sequence or if unresolved isomeric or stereochemical problems remain. The core issue is that purity asks about proportion, while identity asks about chemical correctness, and those are analytically different endpoints.[1][4][6][11]
Is HPLC purity alone enough to evaluate a research peptide?
HPLC purity is valuable, but HPLC purity alone is not the same as full peptide characterization. It generally informs chromatographic impurity burden under a stated method, whereas broader purity assignment and lot qualification may also require identity testing, moisture or solvent information, counter-ion review, and stronger impurity characterization when the project is sensitive to low-level heterogeneity.[1][3][9]
What methods usually provide the strongest peptide identity evidence?
The strongest peptide identity evidence usually comes from at least two orthogonal methods, such as LC-MS combined with relative retention time, peptide mapping, amino acid analysis, or NMR. That approach aligns with both peptide-specific regulatory guidance and pharmacopeial reference-standard practice because it reduces the chance that one method-specific blind spot will drive the conclusion.[1][4][9]
Why do better COAs separate identity, purity, and assay?
Better COAs separate identity, purity, and assay because each line answers a different analytical question. Identity addresses whether the material is the claimed peptide, purity addresses what else is present, and assay addresses quantitative content against a defined method or reference. Combining them into one figure obscures information that laboratories often need for source selection and lot review.[2][4]
What should a research team prioritize when comparing peptide suppliers?
A research team should prioritize lot-specific certificates of analysis, named analytical methods, numerical result values, impurity data, and orthogonal identity evidence where sequence ambiguity or stereochemical risk is relevant. Those elements are more informative than generic purity slogans because they show how the batch was actually characterized and whether the documentation is traceable to the supplied lot.[1][13]
Next Steps
Review batch-specific documentation before selecting any research-use-only peptide. Explore Pure Lab Peptides for RUO peptide compounds with clear labeling, research-focused product information, and available documentation. For research teams comparing peptide suppliers, prioritize COA availability, transparent labeling, and lot-level documentation.
References
- European Medicines Agency. “Guideline on the Development and Manufacture of Synthetic Peptides.” EMA Scientific Guideline. 2025. https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-development-manufacture-synthetic-peptides_en.pdf
- International Council for Harmonisation. “ICH Q2(R2) Guideline on Validation of Analytical Procedures.” ICH / EMA Scientific Guideline. 2023, effective 2024. https://www.ema.europa.eu/en/documents/scientific-guideline/ich-q2r2-guideline-validation-analytical-procedures-step-5-revision-2_en.pdf
- Westwood S, Kaarls R, Churley M, et al. “Methods for the SI-traceable value assignment of the purity of organic compounds.” Pure and Applied Chemistry. 2023. https://doi.org/10.1515/pac-2020-0804
- International Council for Harmonisation. “Q6A: Specifications: Test Procedures and Acceptance Criteria for New Drug Substances and New Drug Products: Chemical Substances.” ICH / EMA Scientific Guideline. 2000. https://www.ema.europa.eu/en/documents/scientific-guideline/ich-q-6-test-procedures-and-acceptance-criteria-new-drug-substances-and-new-drug-products-chemical-substances-step-5_en.pdf
- Badgujar D, Paritala ST, Matre S, Sharma N. “Enantiomeric purity of synthetic therapeutic peptides: A review.” Chirality. 2024. https://doi.org/10.1002/chir.23652
- Petersson P, Buckenmaier S, Euerby MR, Stoll DR. “A Strategy for Assessing Peak Purity of Pharmaceutical Peptides in Reversed-Phase Chromatography Methods using Two-Dimensional Liquid Chromatography Coupled to Mass Spectrometry. Part I: Selection of Columns and Mobile Phases.” Journal of Chromatography A. 2023. https://doi.org/10.1016/j.chroma.2023.463874
- Lian Z, Wang N, Boyne MT 2nd, et al. “Characterization of Synthetic Peptide Therapeutics Using Liquid Chromatography-Mass Spectrometry: Challenges, Solutions, Pitfalls, and Future Perspectives.” Journal of the American Society for Mass Spectrometry. 2021. https://doi.org/10.1021/jasms.0c00479
- Zeng K, Geerlof-Vidavisky I, Gucinski A, Jiang X, Boyne MT 2nd. “Liquid Chromatography-High Resolution Mass Spectrometry for Peptide Drug Quality Control.” The AAPS Journal. 2015. https://doi.org/10.1208/s12248-015-9730-z
- McCarthy D, Bhavsar K, Colon YJ, et al. “Reference Standards to Support Quality of Synthetic Peptide Therapeutics.” Pharmaceutical Research. 2023. https://doi.org/10.1007/s11095-023-03493-1
- U.S. Food and Drug Administration. “Guidance for Industry: Synthetic Peptides.” FDA Guidance Document. 2017. https://www.fda.gov/media/107622/download
- Verbeke F, Wynendaele E, Braet S, D’Hondt M, De Spiegeleer B. “Quality evaluation of synthetic quorum sensing peptides used in R&D.” Journal of Pharmaceutical Analysis. 2015. https://doi.org/10.1016/j.jpha.2014.12.002
- Currier JR, Galley LM, Wenschuh H, et al. “Peptide impurities in commercial synthetic peptides and their implications for vaccine trial assessment.” Clinical and Vaccine Immunology. 2008. https://doi.org/10.1128/CVI.00284-07
- International Council for Harmonisation. “Q7 Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients.” ICH / European Commission Guideline. 2000. https://health.ec.europa.eu/document/download/e2703770-5bef-492e-8d43-1f579d12a475_en?filename=q7astep2_en.pdf











